Farmaindustria explains to Esclerosis Múltiple Euskadi what these studies consist of, how to take part in them and how research into this pathology is progressing
Source: farmaindustria.es
The involvement of patients in the entire process of research, development and approval of new medicines is a need that is increasingly accepted by all actors in the healthcare system. But in order to make it as enriching as possible, it is necessary for these patients to know first-hand what the process consists of and how they can contribute to improving it.
In this context, the Basque Multiple Sclerosis patients’ association held a webinar last Tuesday dedicated to what patients should know about clinical trials. In it, Amelia Martín Uranga, associate director of Clinical and Translational Research at Farmaindustria, underlined the importance of biomedical research for modern healthcare systems due to its value both for patients – for whom it opens up new hope for treatment – and for centres – as it means savings in medicines – and healthcare professionals, who are at the forefront of scientific knowledge.
He also pointed out that clinical trials require a large investment by their promoters, who are mainly pharmaceutical companies: in the case of Spain, these are responsible for eight out of every ten trials launched, Martín Uranga recalled. And, above all, he stressed that this is a long and complex process, as the pharmaceutical industry spends an average of 10-12 years developing and bringing a new drug or vaccine to the patient, and only seven out of every 100 drugs that make it to the clinical phase end up being marketed.
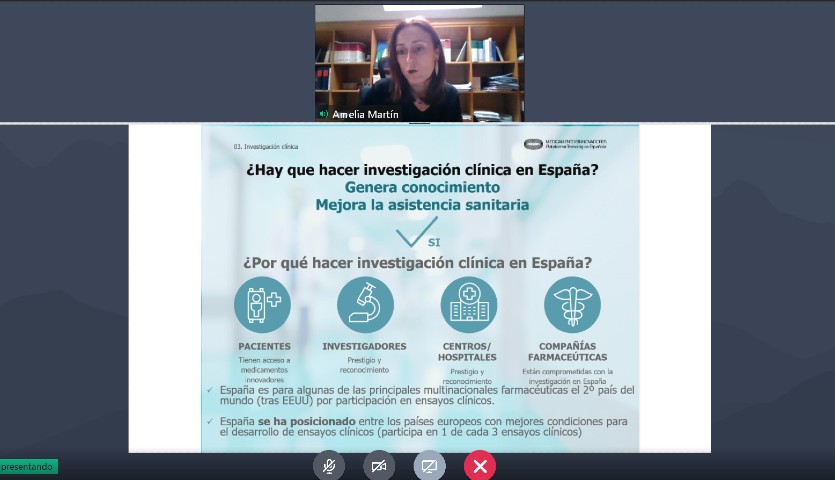
Spain has become a benchmark country in clinical research, as confirmed by the European leadership in clinical trials for Covid-19. But it is not the only example: in multiple sclerosis, more than thirty studies have been launched in Spain alone in the last three years – at the height of the pandemic – with the participation of 250 centres, with the opportunities that this implies for these patients.
In fact, this pathology is one of the most recent success stories in biomedical research, as in three decades it has gone from a single available drug to fifteen. Moreover, these treatments have changed the course of the disease and significantly improved the quality of life of these patients and their families, although there is still much progress to be made in research, as the Farmaindustria spokeswoman concluded.

