Data from the Spanish Clinical Trials Register reveal the commitment of the pharmaceutical industry, which finances 88% of trials with paediatric patients.
Incentives for innovation are fundamental in this field, along with that of rare diseases, so the new European regulation is a great opportunity to encourage even more research and investment that can benefit European paediatric patients.
Farmaindustria participates in the annual meeting of the Spanish Network of Paediatric Clinical Trials
Source: farmaindustria.es
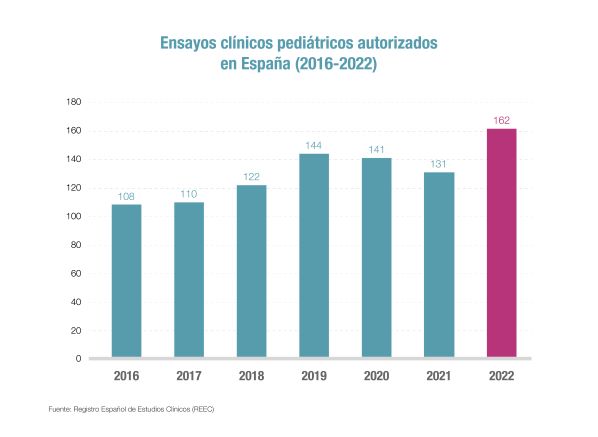
Clinical trials in the paediatric population have been increasing since 2016 to reach a record 162 authorisations last year, according to data from the Spanish Clinical Trials Register (REEC), which are not yet definitive and could increase. The figure is 24% more than in 2021, when, despite more trials being authorised overall, the number of paediatric trials was 131.
The trend is upward and shows the commitment of pharmaceutical companies to the paediatric population, as they drive 88% of the studies (in the total number of trials, this sector finances 86%). This has enabled Spain to position itself as an international leader in clinical trials in recent years, as evidenced by the record investment in R&D in 2021: 1,267 million euros, 789 million of which are dedicated to clinical trials.
This was explained by the associate director of Clinical and Translational Research at Farmaindustria, Amelia Martín Uranga, at the annual meeting of the Spanish Network of Paediatric Clinical Trials (Reclip), on Paediatric clinical research in the national and international context: advances and challenges.
By pathology, cancer accounted for most paediatric trials (15.9%), followed by nervous system pathologies (10%). Congenital, hereditary and neonatal abnormalities, haematology, viral diseases, immune system pathologies and respiratory diseases are the next most authorised trials last year.
Of the total number of paediatric clinical trials, 43% are already in phase I and II, the so-called early phases of research. “This figure is increasing and is important because it means that patients can benefit from the effects of new drugs from the earliest stages of research,” said the Farmaindustria spokeswoman.
In 2021, Farmaindustria published the latest update of the Guide to Clinical Research in Early Stages, a model that makes it easier for researchers, patients and pharmaceutical companies to find the reference centres that exist in Spain for research in these stages. Spain currently has 17 early clinical research units that admit paediatric patients, out of a total of 39.
Farmaindustria’s Director of Clinical and Translational Research spoke about the European Pharmaceutical Strategy, one of whose main initiatives is the reform of both the sector’s general legislation and Regulation 1901/2006 on paediatric medicines: “Together with that on orphan drugs, this regulation is a success story, and taking into account that rare diseases and paediatric diseases are among the most complex, the European Commission should promote additional incentives to attract investment in this field”.
Martín Uranga also analysed the adaptation to the new European regulation on clinical trials: “With the new regulation, competition between countries will increase and, in order to maintain the leadership achieved by Spain in this field, it is important to reinforce the structures and technical and human resources that allow for a correct adaptation to the new European regulations on clinical trials”.
In this regard, the Farmaindustria spokesperson highlighted the need to strengthen public-private collaboration through an ecosystem of alliances and collaborative networks that currently exist at national (Reclip) and pan-European (IMI-Conect4children) levels, so that public and private institutions can jointly address the scientific and regulatory challenges posed by this type of trials for children. or the project that promotes cross-border trials within the EU (Borders should no longer be barriers). She also addressed the importance of explaining to society the value of the European Health Data Space and of sharing health data with the ethical and legal guarantees promoted by the pharmaceutical industry.
The associate director of Clinical and Translational Research of Farmaindustria shared a colloquium on The European Regulation on Clinical Trials and its impact on paediatrics with the head of the Paediatric Oncology and Haematology Department of the Vall d’Hebron Hospital, Lucas Moreno, and the head of the Clinical Evaluation and Pharmacology Division of the Spanish Agency for Medicines and Health Products (Aemps), María Jesús Fernández Cortizo.

