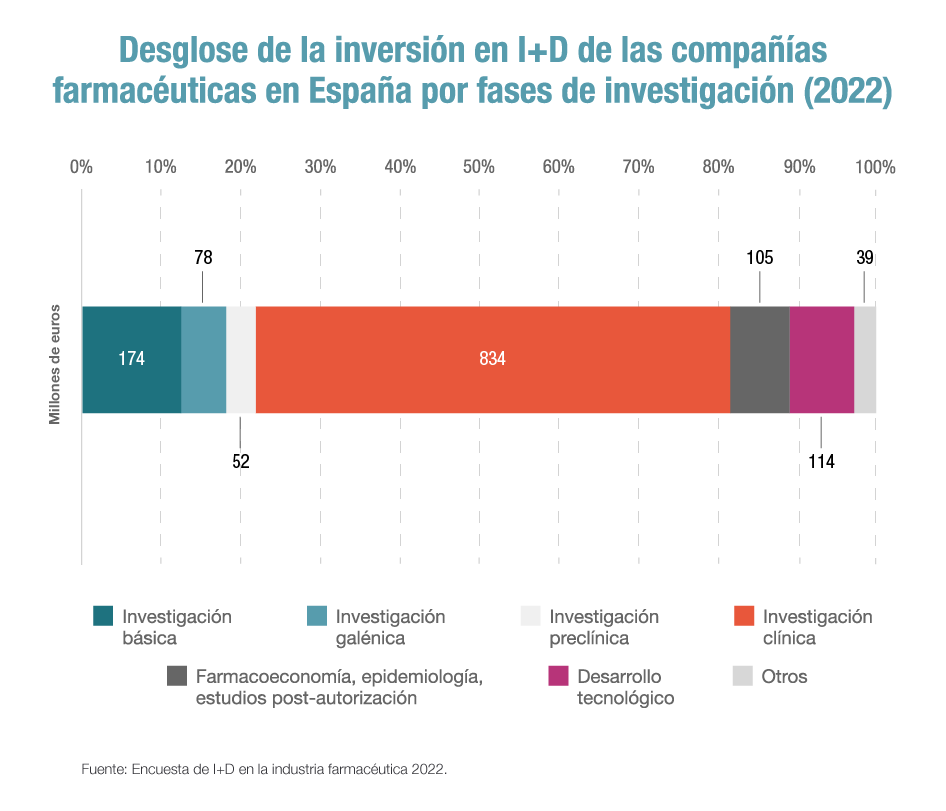
Of the €1.395 billion registered in 2022, 60% was devoted to clinical trial development.
One out of every eight euros is already spent on basic and pre-clinical research, the most complex and highest risk area.
“Patients benefit most from this investment, because a large part of it is dedicated to launching clinical trials, a unique opportunity to cure diseases,” says director general Juan Yermo.

farmaindustria.es
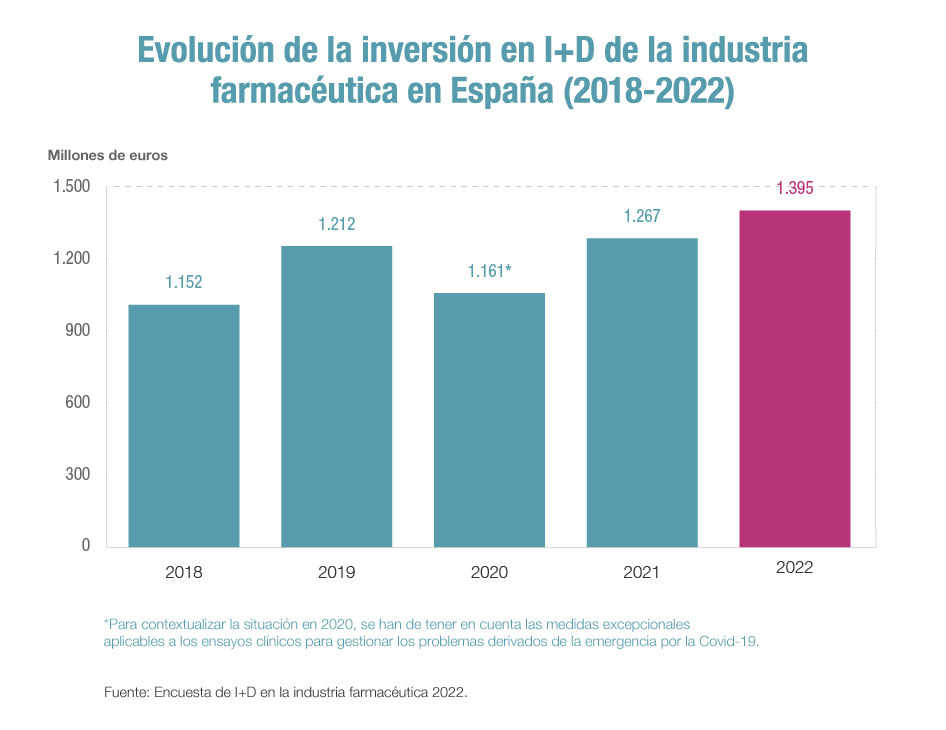
Investment in drug research and development (R&D) by the innovative pharmaceutical industry reached a new all-time high in 2022 in Spain by allocating €1,395 million to these activities, 10.1% more than in 2021 and the largest increase in the last 15 years in this item.
This is reflected in the latest survey on R&D in the pharmaceutical industry 2022, which Farmaindustria conducts each year among its member companies, which also notes that 60% of the nearly 1,400 million euros mentioned were devoted to developing clinical trials – 5.6% more than the previous year and the item that represents the most investment within the different phases of research into new treatments.
Companies’ commitment to clinical research in Spain has increased at an average annual cumulative rate of 5.7% over the last 10 years, rising from 479 million euros in 2012 to 834 million euros in 2022. For many pharmaceutical companies, Spain is already the second country in the world in terms of clinical trial activity, behind only the United States, and participates in one out of every three trials launched in Europe.
Similarly, the pharmaceutical industry invested 175 million euros in basic research. This is the second item with the largest allocation of resources, which has experienced a growth of more than 11% compared to 2021 and means that one out of every eight euros of biomedical R&D is devoted to these basic and preclinical phases, the most complex. This is followed by 114 million for technological development and another 105 million for pharmacoeconomics, epidemiology and post-authorisation studies.
“We are proud that the amount of investment in R&D is growing every year, which has made us the industrial sector that invests most in research in our country, both in absolute terms (together with the automotive sector) and in relative terms (in relation to our size or turnover), where we also lead the ranking, in this case together with the aerospace sector. Patients benefit above all from this investment, because a large part of it is dedicated to launching clinical trials, which means that thousands of people in Spain are gaining early access to the treatments of the future, to that new drug, that new innovative therapy for their disease, which, if all goes well, will see the light of day four or five years later. And this, for many patients with serious diseases, represents a unique opportunity to cure their illness,” says Farmaindustria’s director general, Juan Yermo.
This commitment to clinical research also allows more and more Spanish hospitals to participate in trials with new drugs, which “helps our healthcare professionals to be at the forefront of scientific knowledge and to improve the quality of our healthcare system,” adds Yermo.
Collaboration with hospitals, universities and research centres
The pharmaceutical industry allocated almost half of the investment in Spain, 633 million euros, to research contracts with hospitals, universities and public and private centres (what the survey calls “extramural investment”). This is a key contribution to ensuring the financial sustainability of these centres, increasing the skills of their researchers and facilitating access to cutting-edge treatments for patients involved in clinical trials.
La magnitud de esta aportación —auténtico paradigma de la colaboración público-privada en nuestro país— ha sido refrendada recientemente por el Instituto Nacional de Estadística (INE) en su estadística sobre actividades de I+D, que sitúa a la industria farmacéutica como el primer sector de la economía española por sus inversiones extramuros, que suponen aproximadamente un tercio del total de todo el sector industrial.
En cuanto a la “inversión intramuros”, la ejecutada en los centros de investigación de las propias compañías, alcanzó los 762 millones de euros, representando el 55% del total de la inversión en I+D.
Según la encuesta, todas las comunidades autónomas captaron inversiones en I+D por parte de la industria farmacéutica innovadora, aunque Madrid (30,4%) y Cataluña (26,7%) acaparan más de la mitad de la inversión total en España. “Necesitamos más centros para hacer investigación clínica, puesto que el 57% de los ensayos se concentran en dos autonomías. La pandemia ha sido clave en este aspecto, porque demostró que con la digitalización se puede lograr esa necesaria descentralización y que cada vez más pacientes tengan cerca la posibilidad de participar en un ensayo clínico. Esto conllevará una mayor equidad en todo el territorio y que las oportunidades no dependan del código postal”, señala el director general de Farmaindustria.
In pursuit of this objective of decentralisation and, therefore, equity, Farmaindustria took a step forward at the end of November with the presentation of the Guide of recommendations of good practices for the promotion of clinical research in Primary Care, which will bring new treatment opportunities closer to patients in health centres.
On the other hand, in recent years the survey has shown how, within the total investment in clinical research, early-stage trials have gained considerable weight, rising almost 10 points from 26.8% in 2012 to 36.4% in 2022 in terms of economic value (in number, phase I and II trials account for 58% of those approved by the Spanish Medicines Agency in 2022). For their part, Phase III clinical trials (those involving a larger number of patients) maintain their weight and the percentage of investment in Phase IV trials (those after the drug has been marketed) falls, although their financial allocation has remained stable in recent years.
“Of course, all this investment in research would not be possible without close collaboration with the health administrations, the Ministry of Health, the Spanish Medicines Agency, the ethics committees for drug research, health and research centres and with professionals and patient associations,” Yermo stresses. “From Farmaindustria we will continue to strengthen this way of working, which has allowed us to position Spain as one of the best countries in the world for clinical research and to turn our country into a true international hub for these clinical trials from which all citizens benefit, we benefit,” he adds.
A growing and increasingly qualified workforce
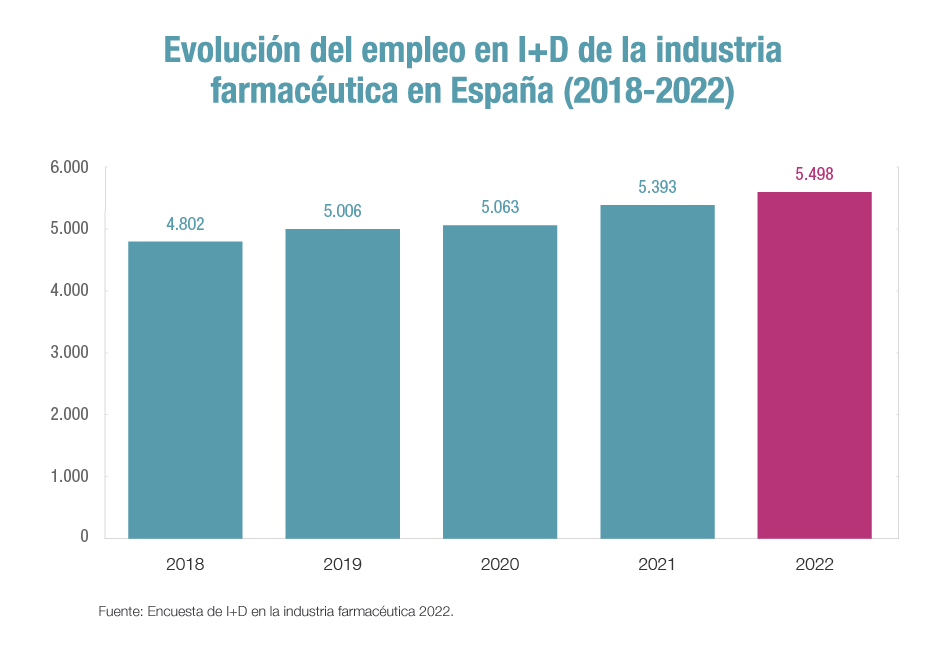
Finally, among the data provided by the R&D in the pharmaceutical industry 2022 survey, the constant growth of direct employment in companies in research departments stands out, which now stands at 5,498 people, the highest figure on record.
This employment, as well as being egalitarian – 67% of the professionals working in R&D departments are women – is highly qualified: in 2022, for the first time, over 90% of university graduates in this area were employed. The qualification of the pharmaceutical industry’s research workforce is one of its most characteristic features. In the last ten years, its share of the total research workforce has increased by almost 9 points.
“The pharmaceutical sector in Spain is a leader in R&D investment, accounting for 20% of all industrial investment in our country; it is an international leader in clinical trials (only behind the United States); it creates high quality, qualified and equal employment; it contributes to national GDP above its weight in employment through its high productivity and its great export capacity, and it is a benchmark in circular economy, social commitment and the SDGs. Moreover, nearly a third of our investment in research is currently allocated to biotechnology, which further reinforces the strategic nature of the sector, provided that the conditions are in place to exploit its full potential. We are in a position to continue investing in this country,” summarises the director general of Farmaindustria.
To achieve this, Yermo proposes reviewing the current support mechanisms for pharmaceutical innovation and public-private collaboration and promoting the digitalisation of the sector, as well as reducing the existing gap with respect to the main European countries in terms of the availability of new treatments and the waiting times for patients to access them. In this way, “we will be able to improve our innovation ecosystem and enhance synergies between national public research and the pharmaceutical industry,” he concludes.

