
Source: elperiodico.com
Multiple sclerosis is an autoimmune disease of the central nervous system that affects the brain and spinal cord. It is caused by damage to the myelin sheath, the protective covering that surrounds nerve cells. When the sheath is damaged, nerve impulses slow down or stop. Among the most common symptoms of the disease are loss of vision, loss of strength in the arms and legs, or a feeling of numbness in the legs. But the disease can vary greatly from person to person, which is why it is known as the “disease of a thousand faces”.
Another symptom that multiple sclerosis can present is dysphagia, difficulty swallowing food. It is one of the least known and was the first one that appeared to Cristina Bajo when she was 20 years old. A misdiagnosis accompanied her until the age of 40, when the first “visible” symptoms began: her right arm and leg became paralysed.
After further tests and studies, she was diagnosed with multiple sclerosis. The delay in diagnosis meant that his disease was in the primary progressive phase, i.e. a phase that is marked by a steady worsening of symptoms without definite recurrences or periods of remission. Years ago, multiple sclerosis was considered a rare disease both because it affected 1 in every 2,000 citizens and because of the lack of public awareness. Currently, according to data from the Spanish Society of Neurology (SEN), 2,000 cases are diagnosed each year and 55,000 people suffer from it in Spain.

Cristina’s story
“The disease was progressing rapidly and I was living in absolute panic because I woke up in the morning and every day I felt worse. My neurologist suggested I enter a clinical trial because, at that time, there was no treatment for primary progressive multiple sclerosis,” explains Cristina Bajo. She entered the phase II trial, known as double-blind, which means that patients do not know whether they are taking placebo or medication. She later found out that she was on medication the whole time. The positive results led to the next phase and everyone in the trial was switched to medication.
Today, after 10 years of research, the drug has been marketed and the trial is in phase IV to study whether it has long-term side effects. The goal of the treatment is to slow progression. “Participating in a clinical trial has changed my life. If I hadn’t taken part, I would probably be bedridden now. A year after entering the trial, the disease slowed down.
Thanks to clinical research, medical knowledge is advanced by studying people. Clinical trials determine how new treatments work in four phases. Last year, in Spain, more than a third of the trials (328) focused on drugs to treat cancer, the area with the most studies, followed by diseases of the nervous system, the area in which multiple sclerosis is included, and pathologies of the immune system. Respiratory, haematological, viral and cardiovascular diseases were next in number of trials in 2022, according to data from the Spanish Clinical Trials Register (REEC). And 25% of clinical trials are related to rare diseases with 230.
The importance of participating in a clinical trial
Cristina knows that anyone in her situation would have done the same. “When there is medication, it is a choice between medication and trial, but in my case it was either trial or I have nothing to give you”. Although Cristina had the full support of those around her, she admits that she had to make the decision herself. “These are absolutely personal decisions, you are the one who knows what you are facing, how you are, how you feel and what you want from life.
Spain authorised more than 900 clinical trials with medicines last year. The figure is higher than that recorded in 2018 and 2019, years before the pandemic when 800 and 833 clinical trials were authorised, respectively. Eighty-six per cent of the trials were driven by pharmaceutical companies, whose investment in this item has been increasing in recent years to €789 million, 60% of total R&D investment in the sector in Spain, as reflected in the latest R&D Activities Survey published by Farmaindustria.
When you decide to enter a clinical trial, doctors inform you of the pros and cons and, although there is always some fear, Cristina admits that “medicine is an empirical science, you learn by trying, so there is no choice but to do clinical trials and participate in them, it is the only way for it to evolve”.
Investing in research
Clinical research generates a great virtuous circle, as it involves investment in hospitals by the companies promoting the trials; it contributes to the qualification of healthcare professionals, thereby increasing the quality of our healthcare system; and it opens up new possibilities for Spanish patients, for many of whom participation in a clinical trial may represent a unique opportunity to cure their disease.
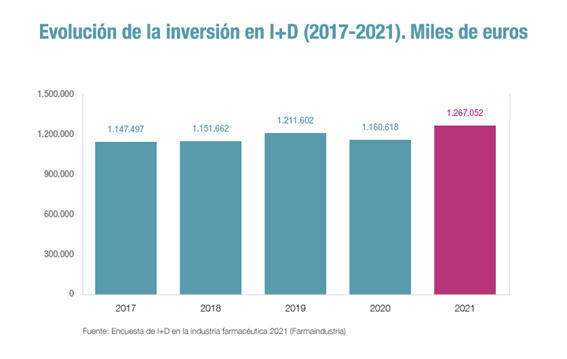
Of the 906 trials published by REEC in 2022, more than half (525) are in the early phases of research, which are considered the most complex in clinical trials and whose promotion represents a challenge in Spain, once its leadership in later phases has been consolidated. Javier Urzay, Deputy Director General of Farmaindustria, points out that “Spain has become a world leader in clinical trials in recent years, thanks to the commitment of the pharmaceutical industry, which finances and promotes more than 86% of the studies, but also thanks to the strength of the health system, the high qualification of its professionals, pioneering legislation and patients who are increasingly involved”.
Thanks to patients like Cristina Bajo, who decided to participate in a clinical trial, the drug is now approved. “I am very happy that others can benefit. For me, it is an ethical obligation for everyone to collaborate with science,” she concludes.

