Our country is establishing itself as a hub for clinical research, thanks to its public healthcare system: in 2025 alone, more than 900 trials were authorised
 Silvia is taking part in a phase III trial, which aims to test the effectiveness of a drug in preventing metastasis in triple-negative breast cancer / eldiario.es
Silvia is taking part in a phase III trial, which aims to test the effectiveness of a drug in preventing metastasis in triple-negative breast cancer / eldiario.es
Silvia was diagnosed with triple-negative breast cancer fifteen months ago. This subtype accounts for around 10% of all tumours found in this organ. It usually affects young women—she was 50—and is quite aggressive. If detected early, with the standard treatment of chemotherapy and immunotherapy to reduce the tumour as much as possible, and surgery to remove the breast, the five-year survival rate is well over 70%. However, if, after the operation, the removed tissue still contains traces of tumour cells, the prognosis is bleaker.
An international group is testing a combination of innovative drugs on these patients to see whether their use can prevent the development of metastases. “My oncologist suggested I take part in a clinical trial and I agreed to everything. My cancer is one of the worst kinds and I have complete faith in the doctors. Without research, we’re lost,” explains Silvia.
This trial, in which Gregorio Marañón Hospital is participating, involves around 1,500 women worldwide. “Fortunately, there aren’t many, but we want to cure them all,” explains oncologist Sara López Tarruella. This centre is one of the leading institutions for cancer clinical trials in Spain, but the country has become a benchmark at European level and is one of the preferred destinations for laboratories on an international scale.
In 2025 alone, the Spanish Agency for Medicines and Health Products (AEMPS) authorised 962 clinical trials involving human participants. “We are leaders in clinical trials in Europe. We have taken these trials to hospitals across the country,” boasted Prime Minister Pedro Sánchez on Monday during an event to mark the 40th anniversary of the General Health Act. Currently, 2,306 trials are underway.
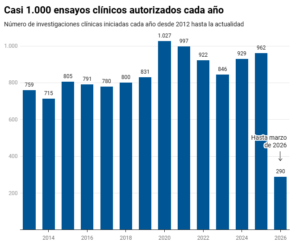 Almost 1,000 clinical trials are authorised each year
Almost 1,000 clinical trials are authorised each year
eldiario.es
“Spain is in a privileged position when it comes to clinical research,” explains Juan Estévez, head of the clinical trials division at the AEMPS. The Spanish ecosystem has strengths on several fronts, but the main pillar is the infrastructure of its National Health System. “Research is embedded within healthcare, which is very positive for the patient and is managed by their own doctor or a doctor who liaises with them,” he explains. This contact helps to recruit volunteers and patients for whom, in many cases, this trial is their last hope.
“If [the person joining the trial] is your patient, it’s easier to get them to take part, because you can draw up a treatment plan, but if they come from other centres, they’re usually highly motivated to explore those alternatives,” explains Dr López Tarruella.
 Silvia is taking part in a phase III trial, which aims to test the effectiveness of a drug in preventing metastasis in triple-negative breast cancer / eldiario.es
Silvia is taking part in a phase III trial, which aims to test the effectiveness of a drug in preventing metastasis in triple-negative breast cancer / eldiario.es
Alongside the surge in clinical trial approvals that began in 2020, Spain has gained prominence in the registration of such projects at European level. “We were pioneers in implementing regulations that have put us on the global map,” says Amelia Martín, director of research at Farmaindustria, the association representing the majority of companies involved in the research, development and marketing of medicines, who notes that, “for many companies, after the United States and China, Spain is the second or third country in terms of clinical research volume”.
As the following chart shows, Spain leads the way in clinical trial registrations in Europe, with 849 studies. Well ahead of France (732), Germany (711) and Italy (593). And, as the Secretary of State for Science, Innovation and Universities, Juan Cruz, noted during the 6th elDiario.es Economic Forum, this country “coordinates or leads one in every two international clinical trials” carried out in Europe.
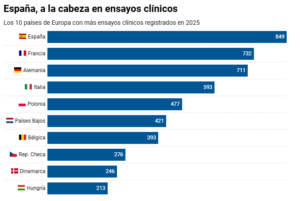 Spain leads the way in clinical trials
Spain leads the way in clinical trials
eldiario.es
For Martín Uranga, one of the keys to this success lies in “the high calibre of our healthcare professionals, an excellent national health system with public hospitals that play a vital role, and supportive, selfless patients who take part in trials that may not work for them, but will benefit those who come after them”.
Although the burden of clinical research falls on the public sector, the bulk of the funding comes from the private sector, which covers 80% of trials through agreements with hospitals, whereby it bears the costs arising from these studies, the recruitment of researchers and the treatment of patients undergoing these trials with innovative and extremely expensive drugs.
“The return that such research provides in terms of learning for healthcare systems regarding new therapies under development is very positive, both for professionals and for patients. And, although people don’t usually like to see medicine and economics mixed together, it must be borne in mind that all activities related to clinical trials are paid for by the sponsor, so patients are participating in a treatment from which they may derive potential health benefits if all goes well,” explains Pau Alcubilla, a specialist in the clinical pharmacology department of the clinical trials unit at Hospital Clínic de Barcelona.
Among the companies with the most clinical trials authorised by the AEMPS in 2025 are Merck, AstraZeneca, Novartis and Pfizer. According to data from the pharmaceutical industry R&D survey, compiled by the sector, companies increased their investment by 40% over five years, reaching €1.775 billion in the last financial year. The budget of the Ministry of Science, Innovation and Universities in 2025 was €17.797 billion.
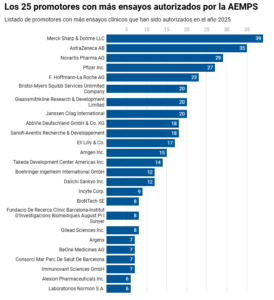 The 25 sponsors with the most clinical trials authorised by the AEMPS
The 25 sponsors with the most clinical trials authorised by the AEMPS
eldiario.es
However, some trials are conducted by hospitals without support from pharmaceutical companies. These are mainly trials that seek to ‘repurpose’ older drugs for new uses or to optimise existing combinations. For example, at the Clínic, where Dr Alcubilla works, many of these academic clinical trials are carried out. “Between 2003 and 2015, the unit was very much focused on supporting optimisation trials in HIV patients, for whom there were more than 30 antiretroviral therapies. We now have a very clear understanding of how to manage them, but 20 years ago it was very important to conduct this type of trial,” he explains.
Currently, most of the clinical trials that are authorised are for cancer treatments. This is no coincidence, nor the result of an impromptu decision. “Twenty years ago, research into genetic biomarkers began, and all that basic research is now reaching the clinical stage,” explains Juan Estévez.
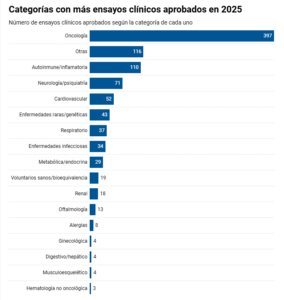 Categories with the most clinical trials approved in 2025
Categories with the most clinical trials approved in 2025
eldiario.es
“In the coming years, we will see precision medicine become increasingly advanced, because breast and lung tumours come in many different forms. Furthermore, we will see new hope for neurodegenerative diseases, which is one of the key areas where significant changes are expected. Or in autoimmune and inflammatory conditions affecting the respiratory system,” predicts the director of research at Farmaindustria.
Ana Martínez is a researcher at the Margarita Salas Biological Research Centre, part of the CSIC. Her team has succeeded in designing a molecule which, in animal models, has corrected one of the most common pathologies in patients with ALS. “We felt it was high time to move on to clinical development and verify that what we see in our models is replicated in patients,” she explains. To do so, she has founded Molefy, a spin-off from the centre, which is majority-funded by the Spanish group Arquimea. “You can go public when you have a repositioned drug, but as soon as there is an innovation, you need the private sector to support you,” admits the scientist.
First-in-human
The drug developed by Martínez and his team received authorisation from the Agency a few weeks ago to begin its journey into Phase I, known as the ‘first in human’ trial. The aim now is to test the safety of this treatment in healthy volunteers, following a series of requirements in the previous phases. Although there are risks, “its safety has been thoroughly tested; we start with very low doses and they are closely monitored,” he explains.
What drives a healthy person to volunteer to be given a drug that has never before been tested on humans? In the case of this trial for ALS, “many people volunteer because a family member has the disease and, in some cases, there is a small financial incentive, as they receive a fee,” he explains. However, money should not be the sole motivation. “It is an incentive, but it should never be the reason for taking part,” adds Alberto Borobia, head of the Central Unit for Clinical Research and Clinical Trials at La Paz Hospital.
Spanish research is a magnet for phase I trials – those involving a few dozen participants, who may be healthy volunteers depending on the condition being studied. This factor underpins the centres’ participation in the subsequent phases: in Phase II, the sample size expands to around 200 or 300 people; in Phase III, the drug’s efficacy begins to be confirmed in thousands of participants; and in Phase IV, long-term studies are conducted once the medicines have been authorised, to monitor for any potential adverse reactions, which are usually rare.
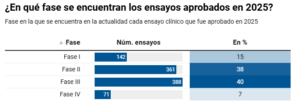 What stage are the trials approved in 2025 at?
What stage are the trials approved in 2025 at?
eldiario.es
Ramón is 29 years old, is completing his clinical pharmacology residency at La Paz Hospital, and has just taken part in one of these phase I trials. Specifically, it was for a vaccine against respiratory syncytial virus in adults which, alongside influenza and SARS-CoV-2, is currently one of the viruses causing high morbidity and mortality in people over 65. “There is a certain risk, but human research is heavily regulated and no clinical trial will go ahead if it is suspected that the risks far outweigh the potential benefits for patients,” he explains.
“I’ve already got my close circle pretty well trained,” jokes Ramón, “but sometimes [Phase I trials] cause a bit of resistance, due to the uncertainty and lack of knowledge, which provoke fear.” He insists he has faith in the research and believes it is “helping to generate more information and enabling the development of a vaccine that can be used on a massive scale among a population that needs it”. The remuneration? He doesn’t deny that it’s good. In his case, around 700 euros for three medical appointments and some blood tests.
 Ramón is a healthy volunteer who has taken part in a Phase I trial for a vaccine against respiratory syncytial virus / eldiario.es
Ramón is a healthy volunteer who has taken part in a Phase I trial for a vaccine against respiratory syncytial virus / eldiario.es
“All Phase I trials involve a high degree of uncertainty, but animal studies and toxicity tests have already been carried out, and there is sufficient evidence to safely begin trials on healthy volunteers,” reassures Dr Borobia. Furthermore, these early phases involve only a small number of volunteers and begin with pairs: one is given a small dose of the drug and the other a placebo. If no risk is perceived, the number of subjects is gradually increased.
Research Ethics Committees play a key role in this process; these are independent bodies that assess the ethical, methodological and legal aspects of the trials. At Gregorio Marañón Hospital, the committee comprises 25 professionals from all specialities, who meet every two weeks. “We review standardised forms, which include everything from the researcher’s CV to their training in good clinical practice and that of their team; we assess multicentre trials, the suitability of the centre, the trial’s objective, the population in which it will be conducted, the budget, the informed consent forms and all the information to be provided to the patient, to ensure their participation is entirely voluntary and they are not coerced into taking part,” explains its chair, María Luisa Navarro.
Areas for improvement: representativeness and access
Despite the progress made and the country’s growing appeal—partly due to its focus on Phase I trial units, improvements to infrastructure, and also the exodus of companies from the UK following Brexit—there is still room for improvement. “The two classic problems in clinical research have been that the results of the trials on which the authorisation of new medicines is based are sometimes not representative of the population receiving those treatments and, at European level, there is concern because a lack of equity in access to clinical trials involving innovative therapies has been identified,” explains Borobia, who coordinates the Readi project —Research in Europe and Diversity Inclusion— from La Paz, a European initiative coordinated by this hospital, involving 73 organisations from 18 countries.
Readi’s aim is to “transform the clinical trial ecosystem by encouraging the participation of under-represented and underserved populations”. This would prevent situations such as the majority of participants in heart failure trials being men aged around 67, when the majority of patients with these conditions are women over the age of 85. Or that socio-cultural factors end up influencing access to innovative therapies.
Accessibility is key, not so much in trials involving healthy volunteers as in phases involving patients who may see their survival rates improve or have the chance of a last-ditch treatment. “Patients from anywhere in Spain can take part, but most clinical trials are conducted in major urban centres. And that is one of the aspects that generates the most debate and calls for improvement: if I live in Madrid, I have more opportunities; if I live in Zamora, I have fewer,” explains the head of the AEMPS. To alleviate this problem, the Agency is drawing up a guide to decentralised elements, which facilitate the digitisation of trials and save on travel. Furthermore, there are programmes and grants to cover certain travel and accommodation costs in some cases.
Catalonia leads the way in terms of the number of centres conducting trials, with a total of 807; followed by Madrid, with 86 centres and 757 trials; and Andalusia, with 64 centres and 522 trials. The trials are not conducted in a single hospital, but are the result of collaborative efforts.
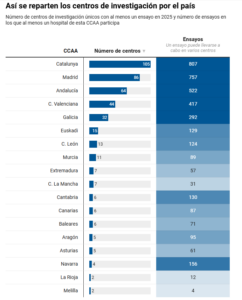 This is how research centres are distributed across the country
This is how research centres are distributed across the country
eldiario.es
Silvia is now halfway through her trial. She adds the empathy shown by the medical team managing her case to the list of the system’s strengths. “Such a humane approach is so necessary in these circumstances,” she says. Her daily life unfolds in that balance between uncertainty and confidence. “If we don’t do it ourselves… whatever can be done to help others, we’ll go for it.”

