
lavanguardia.com
Before finally agreeing to her doctor’s proposal, Cecilia Rodríguez had undergone chemotherapy, radiotherapy and immunotherapy. The cancer she had been living with for six years had spread, and her oncologist told her about an experimental treatment being trialled by a Japanese pharmaceutical company in Barcelona. This marked the start of her first experience as a patient in a clinical trial aimed at reducing the tumour burden of the metastases. So far, it hasn’t grown any further. Although she doesn’t know if the medication will ultimately work, Cecilia says her quality of life has improved and that, in the last six months of being part of this research, she has already noticed “incredible emotional relief”.
Something similar happened to Maria Masferrer (not her real name) when she was asked to take part in a clinical trial, also in the Catalan capital, to test a treatment that could cure the chronic condition she has been living with for some time. “It’s an experimental vaccine. When you have no other option, or the existing ones aren’t satisfactory, you’ve got nothing to lose,” she explains. Over the coming months, she will remain under strict medical supervision to monitor the treatment’s effectiveness: “The risks are minimal and, ultimately, it’s also a selfless contribution to science on my part.”
lavanguardia.com
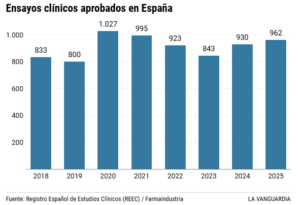
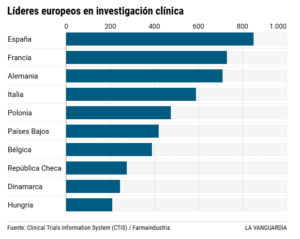
Both Cecilia and Maria are an essential part of the clinical research machinery, a long-term endeavour in which Spain has taken the lead in recent years within Europe. Last year alone, the pharmaceutical industry’s investment in R&D in Spain amounted to nearly €1.7 billion. And the lion’s share of that went to clinical trials. A study by Farmaindustria, based on data from the Spanish Register of Clinical Trials (REEC), confirms that 962 clinical trials involving medicines were authorised in Spain last year. This record figure places the country as the European leader in terms of the number of trials, ahead of France, Germany and Italy. Four out of ten studies were dedicated to finding new drugs to treat cancer, the most commonly treated condition, followed by disorders of the immune, nervous and cardiovascular systems. The association estimates that around 170,000 Spanish patients are currently participating in some form of trial. The economic potential of this activity is clear.
But this leadership – explains Amelia Martín Uranga, Director of Clinical and Translational Research at Farmaindustria – did not emerge overnight. “We were the first country to implement the European regulation on clinical trials. That pioneering regulation was key,” she says. This early regulatory impetus is complemented by other factors, such as a hospital network with almost 1,000 centres involved in clinical trials over the last five years, an ‘altruistic’ patient profile – Spain has one of the highest recruitment rates – and a public-private partnership model in which pharmaceutical companies fund over 80% of these studies.
A clinical trial is experimental research carried out on humans – who may be healthy or ill – to assess the efficacy and safety of new medicines or diagnostic techniques. They are essential for the advancement of medicine and must be approved by ethics committees and the relevant health authorities.
Last year, 962 clinical trials involving medicines were authorised in Spain
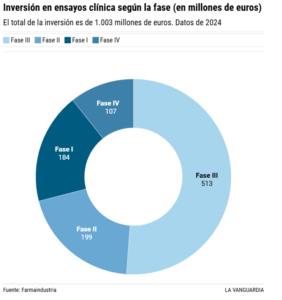
These trials are divided into phases that progress step by step until the medicines reach the market. The first and earliest phase involves small groups of fewer than 100 people to assess the safety of the drug. The second phase increases the number of patients to around a thousand to measure efficacy. The third phase is used to confirm safety and efficacy, determine the optimal dose and assess potential side effects. At this stage, we are dealing with much larger populations of thousands of people, and hospitals from different continents are involved to compare genetic profiles. Finally, the fourth phase involves monitoring once the drug has been marketed.
Spain is home to the hospital that leads the way in clinical research across Europe. The Vall d’Hebron campus in Barcelona — which includes the Vall d’Hebron Research Institute (VHIR) and the Vall d’Hebron Institute of Oncology (VHIO) — hosted more than 2,200 ongoing studies throughout 2025, of which more than 1,800 were clinical trials, explains Dr Xavier Cañas, Director of Commercial and Contracted Clinical Research at the VHIR. Half of these focused solely on the fields of oncology and haematology. “During 2025 we managed more than 2,300 patients, 2,300 people hoping to improve their health,” he notes.
As Dr Josep Comín, Director of Innovation, Research and Universities at the Bellvitge-Viladecans Hospital Management, points out, clinical trials form part of the “mission” of hospitals. “They improve the quality of patient care, help us standardise many processes and provide access to cutting-edge therapeutic innovations,” he notes. This centre has established a dedicated unit that currently has 270 active research projects, involves 161 different sponsors and is conducting trials on around a thousand patients.
lavanguardia.com
At the other end of this innovation chain are the pharmaceutical companies. The vast majority of those choosing Spain to trial their medicines are foreign corporations, and these have also been establishing a presence here with more R&D centres and digital health hubs. Yolanda Martínez, Director of Clinical Operations for the US-based Bristol Myers Squibb (BMS) in Spain and Portugal, points out that Spain is now the company’s second-largest market after the US, both in terms of the number of trials and the number of patients enrolled.
“We are transitioning from being a country that is very good at recruiting patients to generating more and more clinical evidence,” she notes, referring to the growing proportion of early-phase trials (Phases 1 and 2), in which local researchers can play a much more active role in the initial stages of drug development. The group has a translational research centre in Seville dedicated to turning laboratory discoveries into real-world applications for patients.
80% of these studies are funded by the pharmaceutical industry through hospitals and R&D centres
Another of the sector’s giants, the Swiss company Roche, allocated €57.1 million to clinical research in Spain in 2025, an investment that enabled it to launch 283 clinical trials involving 76 different molecules. More than 8,500 patients took part in these trials – in this respect, Spain is second only to the United States and China – along with nearly a thousand healthcare professionals from 178 hospitals. “The vast majority of Roche’s medicines currently received by patients worldwide in areas such as oncohaematology, neurology or ophthalmology have passed through Spain at some point during their research and development phase,” replied Mariluz Amador, head of the medical department at Roche Farma Spain, via email.
Meanwhile, fellow Swiss firm Novartis conducted 255 clinical trials in Spain last year, in collaboration with more than 150 research centres, 2,850 professionals and involving 7,140 patients. “In this drive for innovation, we are focusing on major health challenges such as cancer and cardiovascular diseases, the two leading causes of death in Spain,” say representatives of the multinational pharmaceutical company.
Nevertheless, both the pharmaceutical industry and healthcare centres face some of the key challenges Spain must overcome to improve its position, particularly in the face of fierce competition from the United States and certain countries in the Asia-Pacific region, such as China. “We need to provide more regulatory flexibility to ensure Europe remains attractive. It is worth bearing in mind that there is the economic cost factor, but we must play the historical prestige card to maintain Spain and Catalonia as centres of excellence,” says Pau Alcubilla, a clinical trials physician at Hospital Clínic in Barcelona.

lavanguardia.com
Indeed, Bristol Myers Squibb (BMS), the Spanish Association for Cancer Research (ASEICA) and the Platform of Patient Organisations (POP) presented a report at the end of February in which they warned that Spain could miss out on the economic benefits of clinical trials—estimating a cumulative total of €1.262 billion by 2040 — if it does not strengthen its innovative role in this field. Yolanda Martínez, from BMS, points out that a gap in access to these cutting-edge medicines still persists in Spain, as, despite having narrowed, the wait until they reach the market exceeds 600 days.
For Amelia Martín Uranga of Farmaindustria, coordination between agencies, ethics committees and hospitals has been key to reaching the current stage, but it is necessary to continue reducing the bureaucracy surrounding these processes. “The future European biotechnology law will be very important because it will simplify and streamline the management of contracts between the centre and the sponsor,” she stresses. Xavier Cañas, from Vall d’Hebron, also argues that it is “crucial” to reduce these lead times: “We will carry out our work with the same rigour, but a pharmaceutical company’s decision-making will be different if it takes longer in Europe.” Brussels has stepped up its efforts and has also launched the FAST-EU project to speed up the evaluation of clinical trials by multinationals in the EU.
According to Farmaindustria, around 170,000 Spanish patients are taking part in one of these trials
Regarding the reliance on foreign companies to carry out these trials, Josep Comín of Bellvitge-Viladecas Hospital points out that one of the outstanding challenges is to strengthen links with small local start-ups that wish to validate their molecules in the earliest stages of this entire cycle.
The sector agrees that digitalisation, artificial intelligence and the use of data will set the pace for the coming decades. The field of clinical research, adds Mariluz Amador of Roche, is also advancing ever more rapidly towards personalised medicine: “We have on the horizon a more proactive and efficient healthcare model, where precision diagnosis and molecular profiling will enable each disease to be treated in the most appropriate way for each patient at the right time”. Furthermore, another objective will be to decentralise clinical trials to bring them closer to patients, so that they do not have to travel unnecessarily to take part in them.

